Whitepapers
Interested in all things GMP and analytical services? Be sure to check out our free whitepaper in the spotlight.
Whitepaper: EU GMP - Import of Medicinal Products
Are you a medicinal product manufacturer looking to import your products into the EU in line with the latest EU-GMP import regulations? Be sure to download our free EU GMP guide! Please enter your contact details below, and you will immediately receive your personal copy via email.


Whitepaper: ICH Stability Studies on Pharmaceuticals
Are you up to date on the latest stability testing guidelines and methods? This whitepaper discusses the importance of different methods for testing the stability of pharmaceutical products, stability study guidelines, and other aspects related to the stability of pharmaceutical products.
Please enter your contact details below, and you will immediately receive your personal copy via email.
Keeping it light
Working at Quercus Lab is very special. The entire staff is made up of unique people from different backgrounds and nationalities.
Every day is different and there are many things that define us as a team. For that reason, we would like to share with you some “Fun Facts” about the Quercus team.
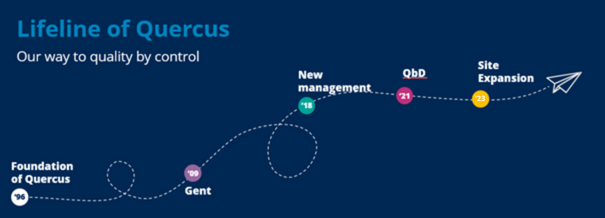
Part of the QbD Group
Supporting life sciences companies form idea to patient
Quercus is part of the QbD Group. The QbD Group supports life science companies worldwide throughout the entire product lifecycle – from idea to patient.
Since 2011, QbD has been providing quality solutions for product development and manufacturing.
The QbD Group’s team offers the skills and expertise for solving complex problems in quality assurance, validation & qualification, regulatory affairs, clinical, quality control, software solutions & services, and go-to-market for companies active in life sciences.
Within our focal points (ATMPs, medical devices, and digital health), we take on a pioneering role on a global level and help determine the rules in these fields.
The QbD Group is headquartered in Belgium and has over 500 consultants worldwide in the Netherlands, Spain, France, the UK, Switzerland, Mexico, Colombia, and the US.