Current EU regulatory framework for MIA holders
In recent years, medicinal products for the EU market are increasingly manufactured outside the EU. Although this trend is particularly evident in the manufacture of active ingredients, it is clear that a similar trend is also occurring in the manufacture of medicinal products.
Moreover, globalization has made supply chains more complex. By classifying importers of medicines as manufacturers and subjecting them to the possession of a manufacturing authorization (MIA), EU regulations already have the basis for a regulatory framework to deal with this situation.
As holders of an MIA, importers are required to comply with good manufacturing practice (GMP) requirements, such as:
- setting up a pharmaceutical quality system,
- having sufficient staff and premises for the activities in question,
- making appropriate arrangements for managing complaints and recalls of medicines,
- and implementing procedures to control their supply chains.
New GMP Annex 21 on August 21, 2022
On August 21, 2022, the new GMP Annex 21 will come into effect and outlines the GMP requirements applicable to Manufacturing Import Authorisation (MIA) holders when importing medicinal products from outside the EU/EEA.
Application
It is interesting to note that this annex applies to
- Medicinal products for human use
- Medicinal products for veterinary use
- Investigational medicinal products
It does not cover products that do not have a marketing authorization in the EU/EEA and are directly re-exported.
Interpretation of the term import
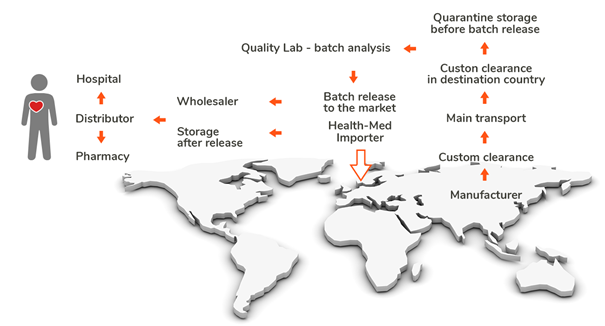
The document provides a common interpretation of the term import, which is defined as the physical introduction of a medicinal product from outside the territory of the EEA/EU. The release of batches of medicines with QP can only take place after physical import and customs clearance in the EU/EEA state.
Sites with specific responsibilities
Sites that are considered to have specific responsibilities with regard to the importation of a medicinal product are:
- Site of Physical Import
- Site of QP certification.
These sites are explicitly mentioned in this Annex 21, but can only be seen in relation to the other actors in the entire supply chain, including
- Manufacturer of the API
- Manufacturer of the medicinal product in the third country
- Transport companies involved
- QC testing site for the imported products
- Marketing Authorization Holder

Where there is no mutual recognition agreement (MRA), it is a legal requirement to test each batch upon import into the EU prior to certification by a competent inspection body and release in accordance with Annex 16 of the GMP Guide.
The requirements for where the testing should be carried out (“in a Member State”) and the minimum testing requirements for products manufactured in third countries are set out in Article 51(1)(b) of Directive 2001/83/EC for medicinal products for human use and in Article 55(1)(b) of Directive 2001/82/EC for medicinal products for veterinary use.
Reference to Eudralex Guidance
The new annex refers to other relevant chapters and annexes in the Eudralex Guidance. The responsibilities related to certification by a QP and batch release were already clearly outlined in Annex 16, but this Annex 21 emphasizes the responsibility of the marketing authorization holder (MAH) throughout the cycle of manufacture and importation of medicinal products.
For example, the guidance states that for the certification of a QP, the marketing authorization holder (MAH) should have access to the complete documentation of the batch at all times.
This is standard practice, but it also points out the key role the MA holder should play in giving the QP access to all necessary documents. The annex explicitly states that the overall responsibility for the marketing of medicinal products rests with the marketing authorization holder. There must be written agreements between the sites involved.
Product Quality Review
There are also some useful clarifications on the Product Quality Review (PQR), which will help the certifying QP appropriately oversee the entire process. As part of this review, the analytical results from the import test site should be compared to those in the certificate of analysis prepared by the third country manufacturer.
The site responsible for certifying the QP must also ensure that there is an ongoing stability program, although the testing may take place in a third country, provided the QP has access to the relevant protocols, results, and stability reports.
Clearly, in an increasingly globalized world, pharmaceutical manufacturing and supply chains can make it complicated to maintain an overview of the entire process.
Why use a contract Lab?
If you want to import medicinal products into the EU, you need an EU import laboratory with QP and GMP accreditation. Setting up such a laboratory is expensive in terms of investment in infrastructure, equipment, personnel, etc.
By outsourcing these activities, you will have a clear picture of the costs upfront and will have access to our experts for QC, QP, and RA for importing medicines into the EU.
Quercus, your trusted GMP Laboratory
Quercus Laboratories is a member of the QbD group and is a trusted GMP Laboratory that supports the pharmaceutical industry with a wide range of QC testing services. We offer a complete service from chemical and microbiological QC testing of raw materials and finished drug products.
Our capabilities include:
Analytical Method Validation – Batch release – EU Import services – Tech transfers of analytical methods – Batch Testing and Certificate of Analysis – Manufacturing plant certification for EU-GMP compliance – Documentary review – Batch Certificate for release – QP Back-Up Services – Additional stability studies – Secure storage of retention samples – MA holder – Regulatory affairs support: CTD dossier writing and marketing authorisation application (CP/DCP/MRP/NP)