The focus now is on Inductively Coupled Plasma – Mass Spectrometry (ICP-MS) and Inductively Coupled Plasma – Optical Emission Spectrometry (ICP-OES).
Scope
Some specific drug products are included/excluded from the elemental impurities limits, as listed below.
Included
- Purified proteins and polypeptides, their derivatives, and products of which they are components (e.g., conjugates)
- Drug products containing synthetically
produced polypeptides, polynucleotides, and oligosaccharides. - All other drug products not specifically excluded
Excluded
- Herbal products (but covered by USP 2322 if they are dietary supplements)
- Radiopharmaceuticals
- Vaccines
- Cell metabolites
- DNA products
- Allergenic extracts
- Cells
- Whole blood
- Cellular blood components or blood derivatives including plasma and plasma derivatives
- Gene-based products
- Cell therapy products
- Tissue engineering (advanced therapy medicinal products)
- Drugs in clinical research phase
The elemental impurities and their toxicity
A total of 24 elemental impurities are listed in the ICH Q3D guideline and have been assigend into 3 different classes based on their toxicity and likelihood of occurence in drug products.
The PDE limits are shown in the table below:
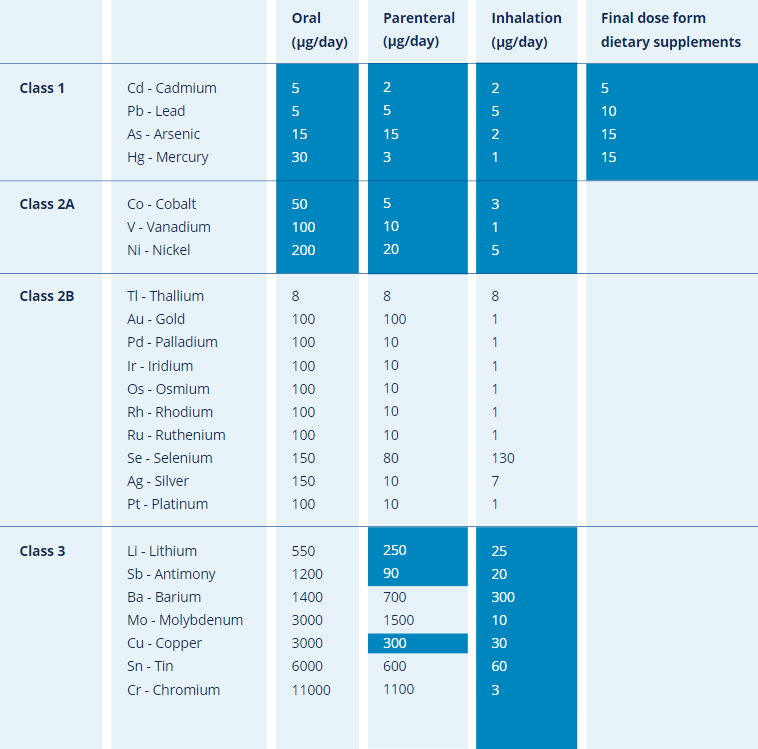
Table – The permitted daily exposure (PDE) limits for elemental impurities in drug products, according to their route of administration. Elements shaded in yellow in the table should be considered in product risk assessment. All elements listed should be included in risk assessment if naturally present or if intentionally or unintentionally added.
Risk-based approach
Elemental impurities may be introduced via APIs or excipients, or they may be introduced during the manufacturing process e.g. catalysts that were added intentionally in synthesis or from with processing equipment or container/closure systems. For the risk assessment, a fishbone diagram is commonly used to ensure all aspects of the process are considered.
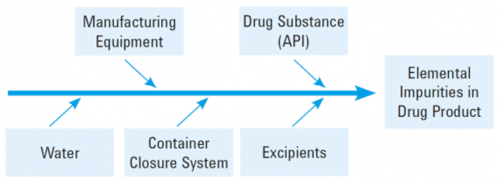
The impact of manufacturing equipment is generally minimal if the equipment and process are well controlled by standard GMP practices, and water can also be eliminated if the pharmacopeial controls currently in place for water for injection or sterilized water are utilized through the manufacturing process.
The drug substance should also be sufficiently controlled through GMP procedures. Container closure system should not pose too many issues in terms of contamination as generally, the levels seen in packaging materials are usually low, though aqueous preparations do need to be considered more thoroughly as this route of transfer from the packaging is a lot more pronounced.
One of the main potential routes of elemental contamination into a drug product is via the excipients. These materials come from a wide variety of sources, and can also be categorized into different levels of risk. Using the information from the risk assessment, elements of concern can be identified.
If an element has been used in the manufacture of or added to the pharmaceutical product, then it must be included as part of the risk assessment. In all other cases, the inclusion of an element into the risk assessment is based on
- the toxicity of the element
- likelihood of presence
- route of administration
The risk assessment results should demonstrate that the elemental impurity levels are consistently less than the control threshold for all lots tested. When conducting the risk assessment, please bear in mind that the limits established, apply throughout the shelf-life of a product.
Control strategy for elemental impurities
The ICH refers to this as a science-based approach that considers all elemental impurities in the same way, regardless of their source. A manufacturer does not need to test for certain impurities if it can be proven that they cannot exist in the final drug product.
Both the ICH and USP identify arsenic, lead, cadmium, and mercury as potential environmental contaminants. Therefore, these four elements must always be included in the risk-based evaluation. If the risk assessment for an existing drug product indicates that all elemental impurities are below 30% of the PDE, no additional control has to be implemented.
A control strategy should be put in place if the risk assessment identifies a source of elemental impurities that is more than 30% of the PDE limit for that drug product. In these cases one should:-
- Identify the source of the Elemental impurity and reduce the level if possible
- Establish a validated method to control the level of Elemental impurity in the drug product.
- In cases where the Elemental impurity level is above 100% of the PDE, one should either stop production and address these high levels, or one can justify the high level of Elemental impurities.
The control strategy needs to be maintained across the product lifecycle, and therefore it is advised to take also samples at the end of shelf-life if testing is required. In this way, also the interaction between the drug and the container-closure system is covered.
In the future, testing will be performed on the drug product, rather than intermediate components, thereby improving patient safety by quantifying the total daily mass of elemental impurities delivered to the patient.
Sample preparation
The sample preparation step of elemental impurities testing must ensure the following:
- put the sample into solution (if it is not already in solution) and stabilize the sample solution, especially if low concentrations of an analyte element are present
- make the analyte concentration fall within the working range of the instrument through dilution or pre-concentration or spiking
- ensure that the sample-containing solution can be nebulized in a reproducible manner.
There are several options for sample preparation and the option you use will depend upon the characteristics of your samples and the instrument technique you are using. It’s worthwhile noting that if your samples are not soluble in aqueous or organic solvents, then you’ll need to use closed vessel digestion, which requires more equipment and expertise.
Analytical Method development and validation
As part of a risk assessment, or as a control strategy, you may need to implement testing for elemental impurities. The ICH Q3D guidelines require that pharmacopoeial procedures are used, or suitable alternative procedures be used.
Typically the analysis will be carried out using ICP-MS as it allows for low detection limits. However, alternative procedures may be used, provided they can be demonstrated to meet the performance requirements defined in the methods.
ICP-MS is a complex technique, but ICP-MS however has much better detection limits than ICP-OES – around 3 orders of magnitude lower for most elements. ICP-MS instruments achieve detection limits in the low ranges, enough to permit accurate determination of all required elements in all dosage forms, including drugs intended for parenteral or inhalation administration, where the PDE levels are typically an order of magnitude lower than for oral medicines.
If your facility is producing a range of products, ICP-MS offers the flexibility to achieve the required limits for all regulated elements in all sample types.

The ICH guidelines and the USP both allow for two different types of validations to take place, namely Limit and Quantitative. A limit test will only show whether a sample is above or below specification. Quantitative validation allows for a numerical result to be generated which may be more useful for monitoring and data gathering as part of a risk-based approach to elemental impurity control. The decision on the extent of the validation will ultimately come down to the needs of the manufacturer and the extent of the testing.

A limit test may be sufficient for a threshold study, but a full validation may provide more robustness if it is to be used for batch release testing. Minimally, the analytical method must meet the following criteria:
- Elements should be at a minimum those required by the ICH/USP
- Linearity is part of system suitability
- Accuracy/precision demonstrated by recovery single preparation at three levels (Typically 25 percent, 100 percent, and 200 percent of the limit. Acceptance criteria for recovery is 70 – 150 percent, for precision, the percent RSD of recovery results from all levels combined is NMT 20 percent.)
- Sensitivity is demonstrated at 25 percent of the limit
Why use a contract Lab?
Investment in the appropriate equipment can be expensive, not only in terms of the cost of the analytical equipment, but also the preparation capability, infrastructure such as gases and extraction, as well as analyst training and qualification of the instrumentation.
The use of an accredited contract analysis laboratory is often a highly effective way of carrying out validations and routine elemental analysis. Benefits include
- Speed of analysis
- Knowledge
- Clear view on costing upfront
Quercus, your trusted GMP Laboratory
Quercus Laboratories is a member of the QbD group and is a trusted GMP Laboratory that supports the pharmaceutical industry with a wide range of QC testing services. We offer a complete service from chemical and microbiological QC testing of raw materials and finished drug products.
Our capabilities include:
Analytical Method Validation – Batch release – EU Import services – Routine QC Batch Analysis – Raw material testing – Microbiological testing – Preservative Efficacy Testing – Stability Studies and Storage – Clean Room Validation – Active Air Sampling – Compressed Gasses Monitoring – Elemental Impurity Testing – Endotoxine Testing – Purified Water Analysis (TOC) – Cleaning Validation
References
- ICH Guideline for Elemental Impurities (ICH Q3D)
- European Pharmacopeial Convention (Ph. Eur.) Heavy Metals Chapter 2.4.8
- USP <231> Heavy Metals and Affected Monographs and General Chapters
- USP<232> Elemental Impurities – Limits
- USP<233> Elemental Impurities – Procedures
- USP<2232> Elemental Contaminants in Dietary Supplements
- ICH Q9 Quality Risk Management
- Q11 Development and Manufacture of Drug Substances


