An antimicrobial or preservative is defined as a chemical agent that either kills or inhibits the growth of microorganisms. Common antimicrobials used in drugs, over-the-counter (OTC) products, and cosmetics include parabens, alcohols, and quaternary ammonium compounds.
Preservative Efficacy Testing (PET) is a microbial challenge method performed to ensure that the preservative in a product, or the antimicrobial action imparted by the product’s properties, is adequate to counteract the introduction of microorganisms. These tests are mandated by both the United States Pharmacopeia (USP) and the European Pharmacopoeia (EP). PET testing should be conducted promptly after the development of a new formulation to confirm the microbiological safety of the product.
Efficacy is assessed by challenging the formulation (in its final packaging, when possible) with a representative range of microorganisms. Samples are taken at predetermined intervals, and the number of surviving organisms is counted. The preservative properties are deemed sufficient if there is either no increase or a significant reduction in the microbial count within a specified timeframe. The exact criteria vary depending on the product type.
The challenge organisms are chosen to represent a broad spectrum of microbial types, including Gram-negative and Gram-positive bacteria, yeasts, and molds. The EP and USP standards slightly differ in the required test organisms for certain pharmaceutical preparations.
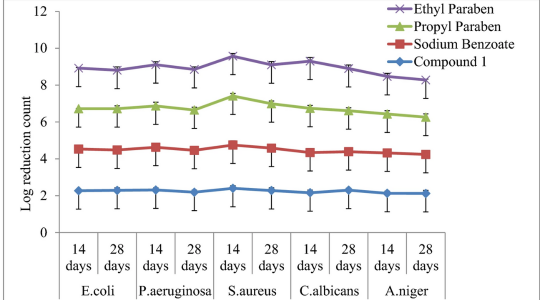
Preservative Efficacy Testing (PET) aids in assessing the microbiological stability of multi-use pharmaceutical products, such as cough syrups and topical creams, which are subjected to repeated dosage withdrawals.
These tests are typically incorporated into stability study protocols and are conducted at both the beginning and end of the study. This approach demonstrates the antimicrobial’s effectiveness throughout the product’s entire shelf life.
For more information about our PET and micro-testing services, please do not hesitate to contact us.


